Materials:Mixtures, Solutions & Colloids Explained
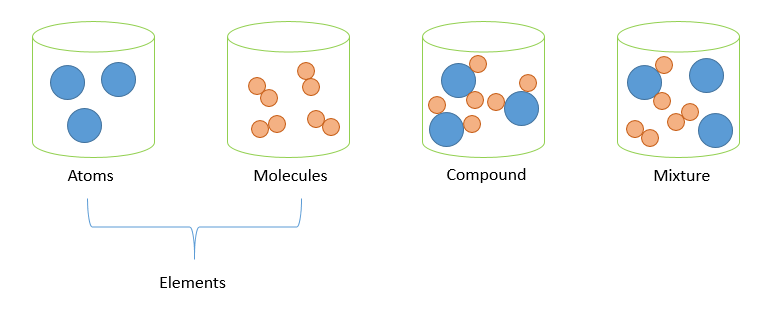
Mixtures
A physical combination of two or more substances.
- Constituents retain their chemical properties
- Can be separated by physical methods
Types of Mixtures
- Solid + Liquid mixture
- Example: sugar in water, salt in oil
- Liquid + Liquid mixture
- Immiscible (do not mix): water + oil
- Miscible (mix uniformly): water + alcohol
- Solid + Solid mixture
- Example: clay and sand, alloys
- Solid + Gas mixture
- Example: smoke, dust, foam mattress
- Liquid + Gas mixture
- Example: fog, mist, clouds
- Gas + Gas mixture
- Example: air
Classes of Mixtures
- Homogeneous mixture → uniform composition (e.g., salt + water, alcohol + water)
- Heterogeneous mixture → non-uniform composition (e.g., sand + water, oil + water)
Differences Between Homogeneous and Heterogeneous Mixtures
| Homogeneous | Heterogeneous |
| Uniform mixture | Non-uniform mixture |
| Constituents cannot be seen | Constituents visible |
| Solute dissolves completely | Solute does not dissolve completely |
| One phase | Two or more phases |
| Stable over time | Constituents separate when undisturbed |
Solute
A substance that dissolves in a solvent.
- Soluble: sugar, salt, ethanol
- Insoluble: sand, chalk, oil
Factors Affecting Dissolution
- Smaller particle size → faster
- Stirring → speeds mixing
- Heating → increases rate
Solvent
The substance in which solutes dissolve.
- Usually present in larger quantity
- Water is called the universal solvent
Examples
| Solvent | Solute |
| Water | Sugar, salt, milk powder |
| Turpentine | Oil paint |
| Petrol | Grease, fat, wax |
| Kerosene | Coal tar |
| Alcohol | Chlorophyll, dye |
| Soap | Fat |
| Benzene | Rubber |
Differences Between Solvent and Solute
| Solvent | Solute |
| Dissolves the solute | Dissolves in solvent |
| Larger quantity | Smaller quantity |
| Usually liquid or gas | Solid, liquid, or gas |
| Lower boiling point | Higher boiling point |
Solution
A homogeneous mixture of two or more substances.
- Solute dissolves completely
- Constituents do not separate
Examples:
- Salt + water
- Sugar + water
- Vinegar + water
- Alcohol + water
Types of Solutions
- Dilute solution → less solute
- Concentrated solution → more solute but can still dissolve more
- Saturated solution → maximum solute dissolved
Suspension
A heterogeneous mixture where solute does not dissolve in the solvent.
- Particles visible to the naked eye
- Particles settle when undisturbed
Examples:
- Gari + water
- Muddy water
- Oil + water
Differences Between Solution and Suspension
| Solution | Suspension |
| Homogeneous | Heterogeneous |
| Particles not visible | Particles visible |
| One phase | Two or more phases |
| Solute dissolves | Solute does not dissolve |
| Cannot be filtered | Can be filtered |
Colloid
A mixture in which one substance is permanently dispersed in another medium.
- Stable, particles do not dissolve
- Too small to see with naked eye
- Appears homogeneous but is heterogeneous
Examples:
- Milk
- Starch in water
- Blood
- Egg
- Toothpaste
Differences Between Colloid and Suspension
| Colloid | Suspension |
| Homogeneous in appearance | Heterogeneous |
| Particles do not settle | Particles settle |